EPKINLY delivered an ORR of 61%, with 38% of patients achieving a deep response of CR1
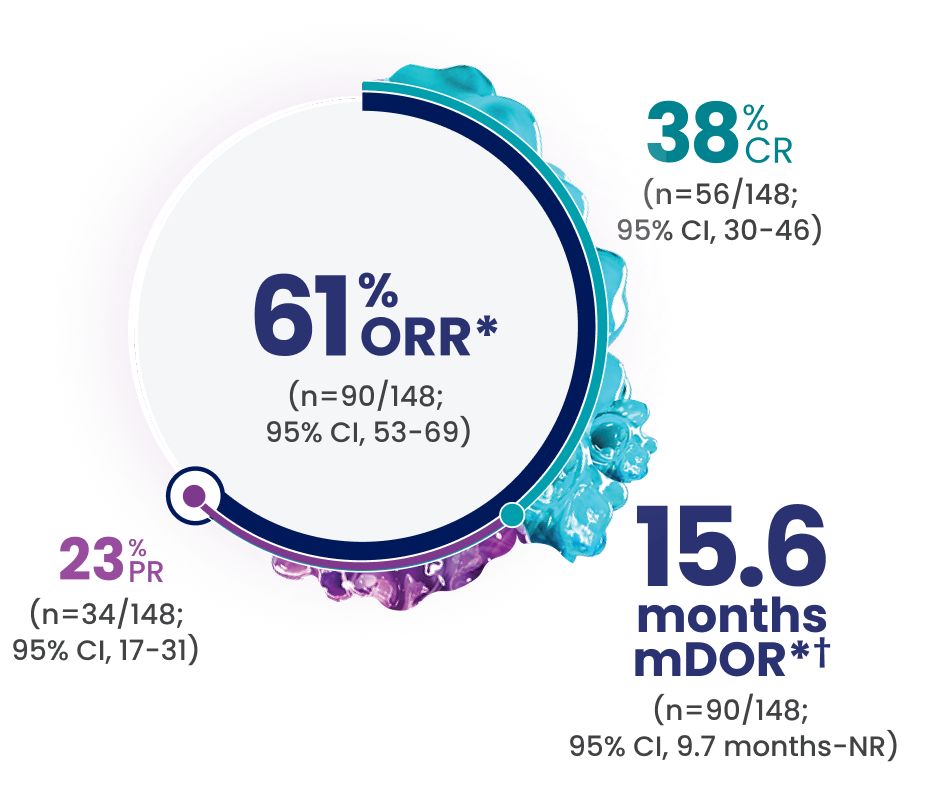
- The median follow-up for DOR was 9.8 months (range: 0-17.3 months)1
- mDOCR*† was NR (n=56/148; 95% CI, 14.3 mo-NR)2
- Complete responses were achieved as late as 10.2 months2
- The median follow-up for DOCR was 9.7 months (range: 8.3-12.1 months)2
- The efficacy of EPKINLY was evaluated in EPCORE® NHL-1, an open-label, multicohort, multicenter, single-arm trial in 148 patients with R/R DLBCL after 2 or more lines of systemic therapy1
Adverse reactions1
- Warnings and precautions include CRS, ICANS, infections, cytopenias, and embryo-fetal toxicity
- Most common (≥20%) adverse reactions were CRS, injection site reactions, fatigue, musculoskeletal pain, fever, diarrhea, COVID-19, rash, and abdominal pain.
- CRS was primarily low grade, predictable, and manageable, with grade 3 (2.5% of patients) and no grade 4 events
- 3.8% discontinued due to ARs; 34% experienced dosage interruptions
Please see more Important Safety Information.
An off-the-shelf subcutaneous
bispecific antibody for DLBCL1
- Enables rapid treatment initiation at the moment of relapse3
- Provide Pneumocystis jirovecii pneumonia prophylaxis during treatment with EPKINLY. Consider providing prophylaxis against herpesvirus during treatment with EPKINLY to prevent herpes simplex and herpes zoster1
NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) Recommended
The NCCN Guidelines® recommend epcoritamab-bysp (EPKINLY) as a NCCN Category 2A treatment option after 2 or more lines of systemic therapy for patients with4‡:
R/R DLBCL NCCN CATEGORY 2A PREFERRED
Histological transformation of indolent lymphoma to DLBCL NCCN CATEGORY 2A RECOMMENDED
NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way.
*Efficacy results determined by Lugano criteria (2014) as assessed by Independent Review Committee (IRC).
†Based on Kaplan-Meier estimate. The median follow-up for DOR was 9.8 months (range: 0-17.3 months) and for DOCR was 9.7 months (range: 8.3-12.1 months).
‡See NCCN Guidelines for the NCCN definitions of Categories of Preference and Categories of Evidence and Consensus.
2L=second line; 3L=third line; AR=adverse reaction; CI=confidence interval; CR=complete response; CRS=cytokine release syndrome; DLBCL=diffuse large B-cell lymphoma; DOR=duration of response; DOCR=duration of complete response; ICANS=immune effector cell-associated neurotoxicity syndrome; mDOCR=median duration of complete response; mDOR=median duration of response; NCCN=National Comprehensive Cancer Network® (NCCN®); NR=not reached; ORR=overall response rate; PR=partial response; R/R=relapsed/refractory.





