
CRS was primarily low grade, predictable, and
manageable1
In patients who received EPKINLY at the recommended 3-step up dosage schedule (n=86)
in the EPCORE® NHL-1 trial
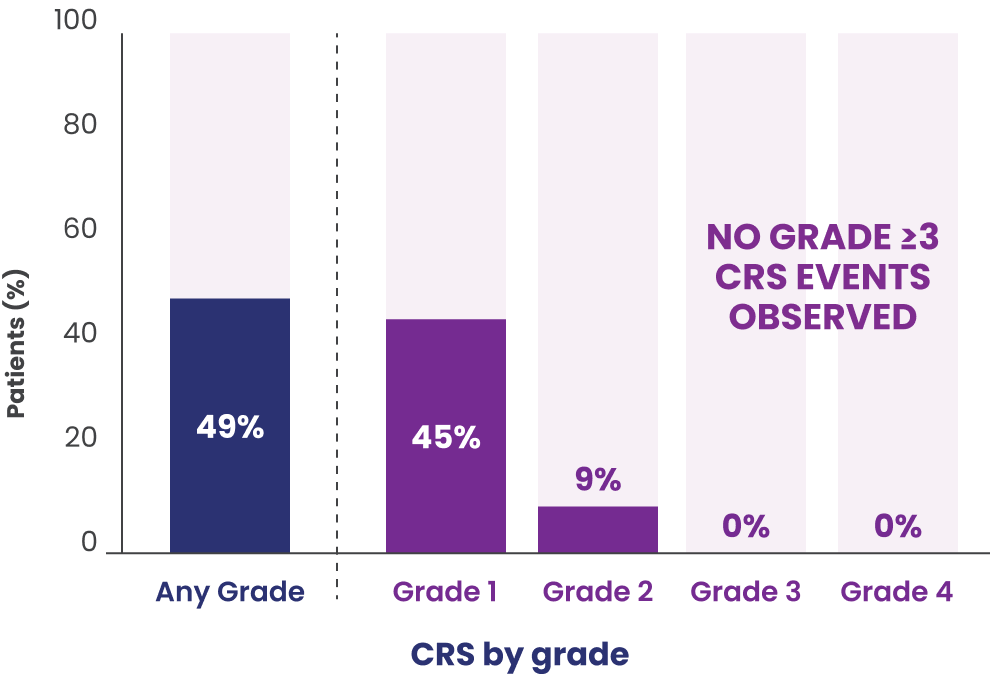
- Recurrent CRS occurred in 48% of patients
- Most CRS events (88%) occurred during cycle 1
- In cycle 1, CRS events occurred in 12% of patients after the 0.16 mg dose, 6% after the 0.8 mg dose, 15% after the 3 mg dose, and 37% after the first 48 mg dose
- The median time to onset of CRS from the most recent EPKINLY dose was 59 hours (range: 0.1-7 days)
- The median time to onset after the first 48 mg dose was 61 hours (range: 0.1-7 days)
Administer pretreatment medications to reduce the risk of CRS and monitor patients for potential CRS. At the first signs or symptoms of CRS, immediately evaluate patients for hospitalization, manage per current practice guidelines, and administer supportive care as appropriate. Withhold or discontinue EPKINLY as based on the severity of CRS. See CRS Management.
SELECT IMPORTANT SAFETY INFORMATION
- For patients with FL, assess whether hospitalization or outpatient monitoring for the first 48 mg dose is appropriate based on comorbidities or other situational factors. During outpatient monitoring after the first 48 mg dose, patients should remain in proximity to a healthcare facility that can assess and manage CRS.
- In patients who experienced CRS, the signs and symptoms included pyrexia, hypotension, hypoxia, dyspnea, chills, and tachycardia. Concurrent neurological adverse reactions associated with CRS occurred in 4.7% of patients with FL receiving EPKINLY monotherapy, and 1.5% of patients receiving EPKINLY in combination with lenalidomide and rituximab (reactions included headache, confusional state, tremors, dizziness, and ataxia).
ICANS events in EPCORE NHL-1 trial in 3L+ FL patients1,2
In patients with FL who received EPKINLY following the 2-step up dosage schedule (n=127)1:
- ICANS events occurred in 6% (8/127) of patients with FL receiving EPKINLY, utilizing the 2-step up dosage schedule
- 3.9% grade 1, 2.4% grade 2
- The median time to onset of ICANS was 22 days (range: 14-66 days) from the start of treatment
- Relative to the most recent administration, the median time to onset was 3 days (range: 0.4-7 days)
- ICANS resolved in 100% of patients
- The median duration of ICANS was 2 days (range: 1-7 days)
- The median duration of exposure for patients was 8 cycles (range: 1-33 cycles)
In patients who received EPKINLY following the recommended 3-step up dosage schedule (n=86)1,2:
- No ICANS events were observed at the time of analysis. The median exposure for patients in the dose optimization cohort was 5 cycles (range: 1-12 cycles). No conclusions regarding the rate of ICANS can be made, as exposure may not be sufficient in this cohort.
Monitor patients for potential ICANS following EPKINLY. At the first signs or symptoms of ICANS, immediately evaluate patient and provide supportive therapy based on severity. Withhold or discontinue EPKINLY per recommendations and consider further management per current practice guidelines. See ICANS Management.
SELECT IMPORTANT SAFETY INFORMATION
- The onset of ICANS can be concurrent with CRS, following resolution of CRS, or in the absence of CRS. Clinical manifestations of ICANS included, but were not limited to, confusional state, lethargy, tremor, dysgraphia, aphasia, and non-convulsive status epilepticus.
3L=third line; CRS=cytokine release syndrome; FL=follicular lymphoma; ICANS=immune effector cell-associated neurotoxicity; mg=milligram; NHL=non-Hodgkin lymphoma.





